Overcoming cancer drug resistance
Researchers at Nano Life Science Institute (WPI-NanoLSI), Kanazawa University, report in Nature Communications how the targeted suppression of lysosome function may lead to brain cancer therapy.
Glioblastoma is a type of brain cancer with a very poor prognosis of survival. Causes of glioblastoma are not known, and there is no method for preventing the cancer. Traditional treatment includes the drug temozolomide (TMZ). In many cases, TMZ kills glioblastoma cells, but a significant portion of patients show resistance to the drug. Changes in the levels of metabolites — small molecules playing key roles in metabolic processes in living organisms — have been observed in TMZ-resistant glioblastoma cells, pointing to the importance of understanding and targeting metabolic pathways in the context of cancer therapy. Now, Atushi Hirao from Kanazawa University and colleagues have investigated the role of lysosomes — cellular organelles that break down biomolecules no longer needed — in metabolic processes linked to the development of glioblastoma. Based on their findings, they propose a targeted lysosomal dysfunction strategy for the treatment of glioblastoma.
Lysosomes perform important biochemical functions. Apart from degrading ‘waste’ biopolymers, they are involved in cell signaling and energy flow and interact with other organelles. As such, they are also relevant for the development and growth of cancer. Hirao and colleagues first checked whether lysosome activity is an indicator of glioblastoma progression. They looked at glioblastoma cell lines taken from patients and found a correlation between lysosomal activity and glioblastoma tumorigenesis and malignancy.
The researchers then investigated how lysosomal activity affects the therapeutic efficacy of TMZ. They found that when administering lysosomal inhibitors, the sensitivity of glioblastoma to TMZ increased, confirming the critical role played by lysosomes in glioblastoma. Further experiments pointed to a protein called transcription factor E3 (TFE3) as a crucial molecule for maintaining lysosomal function, and TMZ tolerance, in glioblastoma cells.
The scientists then studied which amino acids are essential for lysosomal activity in the context of glioblastoma progression. They discovered a correlation between lysine and glioblastoma malignancy. Lysine is not produced by the human body; it must be obtained through nutrition. Hirao and colleagues therefore considered whether a lysine-restricted diet could be a therapeutic strategy for glioblastoma. They pointed out that, while lysine restriction might be a possible approach, it has limited practical use because of toxicity concerns – the human body needs lysine as a precursor to vital proteins.
Instead, the researchers worked out an alternative approach mimicking the effect of lysine restriction. Realizing that one of lysine’s functions is to antagonize the effect of arginine, which plays a role in the biosynthesis of nitric oxide, they tested the use of homoarginine, an antagonist of lysine — that is, inhibiting lysine’s function — to counteract lysine’s blocking of nitric oxide production from arginine, and induce lysosomal dysfunction. Experiments with mice showed that the combination of TMZ and homoarginine led to a significant suppression of glioblastoma cells, demonstrating its potential therapeutic value (Figure).
The work of Hirao and colleagues highlights the critical role of lysosomal function in glioblastoma pathogenesis, and how mimicking lysin restriction may play a role in anticancer strategies. Quoting the researchers: “… disrupting lysosomal function may provide a promising avenue for glioblastoma therapy.”
Background
Glioblastoma
Glioblastoma is a type of brain cancer. It starts with the growth of cells in the brain or spinal cord. The cancer grows quickly and can destroy healthy tissue. Although glioblastoma can happen at all ages, it tends to occur more in older adults. Symptoms include headaches, vomiting, nausea, blurred or double vision, difficulty with speaking, an altered sense of touch, and seizures. There is no cure for glioblastoma; treatments can slow cancer growth and reduce symptoms.
Lysosome
Lysosomes are organelles, enclosed by membranes, located within cells. They contain many types of enzymes (bioproteins with a catalytic function) that assist in breaking down many kinds of biopolymers, such as proteins, carbohydrates, and nucleic acids. As such, lysosomes serve as the ‘digestive system’ of the cell, degrading material from both within and outside of the cell. Lysosomes also serve as signaling hubs, lying at the junction of many metabolic pathways. Recent research has revealed that lysosomes play important roles in cancer development. Detailed understanding of metabolic pathways in the lysosome may help unlock its therapeutic potential. Atsushi Hirao from Kanazawa University and colleagues have now identified a particular lysosome function that is critical for the resistance of glioblastoma cells to the drug temozolomide, a finding that holds promise for anticancer therapies based on lysosome dysfunction.
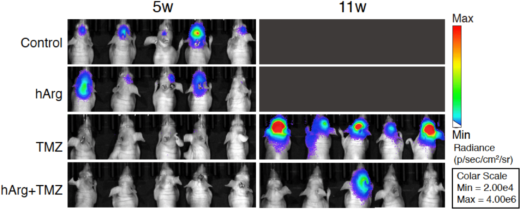
Figure: Glioblastoma tumor growth in mice; comparison of TMZ administration without and with homoarginine.
© 2025 Jing, et al., Nature Communications
Article
- Title
- Lysine-arginine imbalance overcomes therapeutic tolerance governed by the transcription factor E3-lysosome axis in glioblastoma
- Author
- Yongwei Jing, Masahiko Kobayashi, Mahmoud I. Shoulkamy, Meiqi Zhou, Ha Thi Vu, Hiroshi Arakawa, Hemragul Sabit, Sadahiro Iwabuchi, Cong Quang Vu, Atsuko Kasahara, Masaya Ueno, Yuko Tadokoro, Kenta Kurayoshi, Xi Chen, Yuhang Yan, Satoshi Arai, Shinichi Hashimoto, Tomoyoshi Soga, Tomoki Todo, Mitsutoshi Nakada, and Atsushi Hirao
- Journal
- Nature Communications
- Publication date
- Apr 1, 2025
- DOI
- 10.1038/s41467-025-56946-z
- URL
- https://www.nature.com/articles/s41467-025-56946-z

