Multi-state switchable stationary phase opens new doors in chiral separation
Abstract
A team including researchers from Kanazawa University demonstrated HPLC separation of enantiomers using an optically active poly(phenylacetylene) derivative as a chiral stationary phase. The helical conformation of the polymer was altered from a mixed state to left- or right-handed chiral conformations by introducing Na+ or Cs+ ions, respectively, producing a switchable system that offers three different modes of recognition. It is hoped that the system will contribute to the continued development of chiral separation.
Kanazawa, Japan – The concept of chirality can be challenging for the layperson, with “chemical handedness” seeming a very minor distinction. However, as the consequences of the notorious thalidomide disaster illustrate, understanding chiral materials is a major concern. The continued development of chiral separation techniques therefore remains a key research area. A team involving researchers from Kanazawa University has reported a three-state switchable chiral stationary phase (CSP) that provides new opportunities in chiral separation. Their findings are published in the Journal of the American Chemical Society.
Chiral high performance liquid chromatography (HPLC) remains the most effective method for separating chiral molecules. HPLC involves running samples through a tube—known as a column—containing chiral material (the CSP) that can differentiate between pairs of chiral molecules (enantiomers). However, owing to the numerous enantiomers that cannot be separated using currently available CSPs, research in this area remains ongoing.
The researchers report a CSP based on a helical polymer material containing a chiral pendant group that causes the polymer to adopt different conformations in response to metal ions. When Na+ ions are present the polymer is forced to adopt a left-handed helix so that the ions can interact with the aromatic component of the group. In contrast, when Cs+ ions are present the polymer is forced into a right-handed helix to facilitate bonding between the ions and the two oxygen atoms of the group. In the absence of ions the structure is a deactivated mixture of the two helices.
“Our experiments used a CSP based on a poly(phenylacetylene) derivative that can be altered using an achiral external stimulus,” study lead author Daisuke Hirose explains. “This allowed us to control the chirality of the column, and hence the retention of enantiomers, simply by introducing metal salts. The metal ions caused the polymer switch to a particular chiral conformation; however, the effect could be reversed by eluting the column with methanol.”
The stability of the ion-induced states was demonstrated over 4 days of continuous flow through the column, and the separation performance was exhibited by switching between the active and deactivated state numerous times.
“As far as we are aware, our system is the first reported example of a CSP that can be switched between three different recognition states using a stimulus that is not chiral,” study corresponding author Katsuhiro Maeda explains. “We hope to build on our findings to extend the range of enantiomers that can be separated, which we believe will benefit numerous research areas such as drug discovery.”
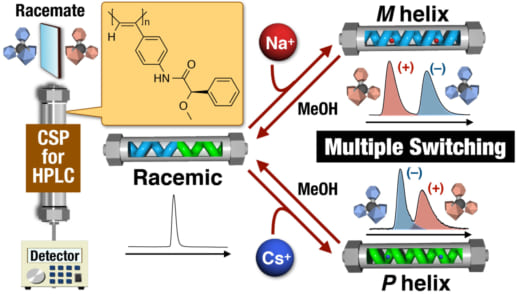
Figure 1.
Schematic illustration of three-state switchable chiral stationary phase based on macromolecular helicity modulation in a poly(phenylacetylene) derivative using metal cations in the column.

Figure 2.
An illustration inspired by three-state switchable chiral stationary phase using metal cations.
Article
- Title
- Three-State Switchable Chiral Stationary Phase Based on Helicity Control of an Optically Active Poly(phenylacetylene) Derivative by Using Metal Cations in the Solid State
- Author
- Daisuke Hirose, Asahi Isobe, Emilio Quiñoá, Félix Freire, Katsuhiro Maeda
- Journal
- Journal of the American Chemical Society
- Publication date
- May 7, 2019
- DOI
- 10.1021/jacs.9b03177
- URL
- https://doi.org/10.1021/jacs.9b03177

